Abstract
BACKGROUND AND PURPOSE: The likelihood of discovering a CSF leak can be determined by assessing intracranial abnormalities. However, the Dobrocky scoring system, which is used to determine this likelihood, did not incorporate patients with CSF-venous fistulas. This study sought to create a new probabilistic scoring system applicable to patients without a spinal longitudinal extradural collection.
MATERIALS AND METHODS: A retrospective review was completed of patients with suspected spontaneous intracranial hypotension who underwent brain MR imaging followed by digital subtraction myelography with same-day CT myelography. Patients with and without leaks found on digital subtraction myelography were included. MRIs were assessed for numerous reported stigmata of spontaneous intracranial hypotension and were compared between cohorts.
RESULTS: One hundred seventy-four patients were included; 113 (64.9%) were women (average age, 52.0 [SD, 14.3] years). A CSF leak was found in 98 (56.3%) patients, nearly all of which (93.9%) were CSF-venous fistulas. Diffuse dural enhancement, internal auditory canals dural enhancement, non-Chiari cerebellar descent, pituitary engorgement, brain sag, dural venous sinus engorgement, and decreased suprasellar cistern size were associated with a CSF leak. A probabilistic scoring system was made in which a single point value was assigned to each of those findings: 0–2 considered low probability and ≥3 considered intermediate-to-high probability of a CSF leak.
CONCLUSIONS: This study offers a new probabilistic scoring system for evaluating the likelihood of discovering a CSF leak on the basis of intracranial MR imaging findings, though the new system is not superior to that of the Dobrocky method for predicting the presence of CSF leaks.
ABBREVIATIONS:
- AUC
- area under the curve
- CTM
- CT myelography
- DSM
- digital subtraction myelography
- IAC
- internal auditory canal
- SIH
- spontaneous intracranial hypotension
- SLEC
- spinal longitudinal extradural fluid collection
Spontaneous intracranial hypotension (SIH) is a condition caused by spinal CSF leaks.1,2 The condition can be extremely debilitating. SIH is classically characterized by orthostatic headaches but can also lead to long-term disability, decreased consciousness, and even coma.3,4 In recent years, much has been learned about the characteristics and frequency of various types of spinal CSF leaks, particularly with the rise of digital subtraction myelography (DSM) and dynamic CT myelography (CTM).5,6 Nevertheless, accurately diagnosing patients with SIH is difficult, and clinicians often rely on radiologic findings.3 Specifically, patients with SIH typically have some combination of intracranial abnormalities best seen on brain MR imaging: dural thickening and enhancement, brain sag, and engorgement of the pituitary and/or dural venous sinuses,7 a set of findings that is explained by physiologic responses to loss of CSF volume according to the Monro-Kellie doctrine.8
Many of the aforementioned imaging findings can mimic those of other pathologies.9 In 2019, Dobrocky et al10 developed a method to determine the likelihood of discovering a spinal CSF leak based on intracranial findings. This probability score, commonly called the “Bern score,” uses 6 imaging findings: pachymeningeal enhancement, venous sinus engorgement, effacement of the suprasellar cistern, the presence of subdural fluid collections, effacement of the prepontine cistern, and decreased mamilllopontine distance. By assigning point values to these findings, the authors created a scoring system to categorize someone as having a high, intermediate, or low probability of a spinal CSF leak.
Undoubtedly, the widely adopted probabilistic scoring method developed by Dobrocky et al10 has had a positive impact on the field of SIH. It created a simplified approach to diagnosing an often underrecognized condition. However, the cohort used for analysis in the study was based on patients with so-called “fast” CSF leaks, which are typically caused by ventral or posterolateral dural tears and associated with spinal longitudinal extradural fluid collections (SLECs). Thus, the study did not include patients for whom a probabilistic scoring system would be most useful, namely patients without a SLEC in whom CSF-venous fistulas are the most common leak type,11 and in rarer cases patients with distal nerve root sleeve tears. Although the scoring system was later demonstrated to stratify the probability of finding a leak on lateral decubitus DSM in a predominantly “slow” leak cohort,11 the original scoring system development did not include any patients with fistulas.
The current study set out to create a new probabilistic scoring system of intracranial findings that can determine whether a patient suspected of having SIH but having no SLEC on spine MR imaging will ultimately have a spinal CSF leak identified. Thus, we assessed brain MR images in patients with clinically suspected SIH and compared findings between patients with and without a spinal CSF leak.
MATERIALS AND METHODS
Patient Cohort
This study was performed following exemption by the local institutional review board. A retrospective review was completed of consecutive patients with suspected SIH who underwent DSM between December 30, 2021, and November 30, 2022. All included patients underwent lateral decubitus DSM. Patients were excluded if they did not have a pre-DSM brain MR imaging or if images were of poor quality or substantially degraded by artifacts. Patients were also excluded if an SLEC was observed on pre-DSM spinal MR imaging (per our institution's diagnostic algorithm, no patients with an SLEC underwent DSM imaging). For any patients who had undergone a previous DSM (ie, before the study inclusion date), either the first DSM performed that identified a CSF leak or the first DSM performed at our institution (in patients in whom a CSF leak was not identified) and the most recent brain MR imaging preceding the DSM were used for analysis. The number of days between the pre-DSM brain MR imaging and the analyzed DSM was recorded. When we used these criteria, dates for all reviewed DSM examinations ranged from December 21, 2018, to November 15, 2022.
DSM Technique
All DSMs were performed during 2 days, the first with the patient in the right lateral decubitus position and the second with the patient in the left lateral decubitus position.6 Imaging was completed with the patient placed on a wedge-shaped cushion on a tiltable table to position the patient's hips superior to his or her shoulders. After placement of a 20-ga spinal needle into the thecal sac (typically at L2–3 or L3–4), 2 separate DSM acquisitions were performed per side: the first with the flat panel detector focused over the upper spine and the second focused over the lower spine. A total of 11 mL of intrathecal Omnipaque 300 (GE Healthcare) was administered for both runs. Following completion of DSM imaging, the patient was kept in lateral decubitus positioning and transferred to CT for a subsequent entire spine CTM. DSM and CTM images were interpreted as part of a single examination.
Brain MR Imaging Protocol
MR imaging was performed on either a 1.5T or 3T scanner. Nearly all imaging analyses were based on fat-saturated postcontrast 3D T1 sampling perfection with application-optimized contrasts by using different flip angle evolutions (SPACE; Siemens) sequences (TR = 600 ms, TE = 7.2 ms, flip angle = 120°, section thickness = 1 mm, FOV = 250 × 250 mm2). Evaluation of superficial siderosis was based on SWI sequences (TR = 49 ms, TE = 40 ms, flip angle = 15°, section thickness = 3 mm, FOV = 201 × 240 mm2). Assessment of subdural fluid collections was performed by comparing axial postcontrast T1 SPACE sequences with axial T2 FLAIR images (TR = 9000 ms, TE = 149 ms, flip angle = 180°, section thickness = 4 mm, FOV = 220 × 220 mm2).
Image Analysis
Four neuroradiologists reviewed the DSM and pre-DSM brain MR images, with the entire cohort split evenly among the reviewers. Thus, each patient in the cohort was reviewed by only a single radiologist, who reviewed the MRIs, DSMs, and CTMs. The reviewers were blinded to clinical information but not blinded to the official reports or annotations in the PACS system.
Each reviewer individually evaluated all DSMs for the presence or absence of an identified CSF leak (specified as positive, negative, or indeterminate), laterality of the leak, and location of the leak (ie, spinal segment). Leaks were further stratified by subtype, according to a previously validated categorization: type 1 (ventral dural tear with an SLEC), type 2 (posterolateral dural tear/nerve root sleeve tear with an SLEC), type 3 (CSF-venous fistula), type 4 (distal nerve root sleeve tear), and type 5 (other).12
MR imaging brain scans were assessed for multiple potential stigmata of SIH: smooth dural enhancement, smooth dural enhancement specifically involving the internal auditory canals (IACs), subdural fluid collections, superficial siderosis, pituitary engorgement, non-Chiari cerebellar tonsillar descent of >5 mm (meaning tonsillar descent without pointed or “peglike” morphology of the tonsils), dural venous sinus engorgement, “layered” hyperostosis of the calvaria, or descent of the cerebral aqueduct iter below the incisural line.
The definition of layered hyperostosis was based on prior studies in which a secondary layer of bone was seen subjacent to the primary calvarial structure.13,14 Pituitary engorgement was considered positive if the superior margin of the gland was convex on sagittal images. The presence or absence of dural venous sinus engorgement was based on assessment of the midportion of the dominant transverse sinus on sagittal images for convexity of the inferior border of the sinus wall.15 Given the ambiguity of this finding, results were categorized as positive, negative, or indeterminate. Multiple measurements were made on fat-saturated postcontrast sagittal images (or the available T1-weighted image if contrast was not used for the examination, typically sagittal MPRAGE) of the following: mamillopontine distance, suprasellar cistern height (measured between the inferior aspect of the optic chiasm and the superior border of the pituitary), and prepontine cistern width.
Statistical Analysis
All statistical calculations were performed using BlueSky Statistics software (BlueSky Statistics). Means and SDs were calculated for all continuous variables. A χ2 test was used to assess statistically significant differences among categoric variables, and the Student t test was used for differences among continuous variables. To obtain a cutoff for any continuous variables that were significantly associated with CSF leaks (in this case, only the suprasellar cistern measurement), we created a receiver operating characteristic curve using a logistic regression model to select the optimal cutoff. These continuous variables were then converted to binary variables on the basis of their relationship to the cutoff point. For all calculations, the threshold for statistical significance was set to P = .05.
To create a scoring system, we put any variables that were found to be significantly associated with the presence of a CSF leak into a multivariable analysis with a logistic regression. Following the methodology of the prior study,10 any variable that could not be assessed (in this case, dural enhancement in 8 patients who did not undergo contrast-enhanced MR imaging) was counted as negative. Any variables that had a coefficient of >2 were planned to be given a score of 2 for the final system (though none were noted in our analysis); all others were given a score of 1. The scores of all patients were then calculated, and a final logistic regression with the Youden index was performed to assess the optimal cut-point for a scoring system.
Logistic regression analyses were used to assess correlations between a CSF leak and both the scoring system developed in the current study and that of Dobrocky et al.10 Areas under curve (AUCs) were compared between both scoring systems.
RESULTS
Patient Cohort and CSF Leaks
Of 175 patients, one was excluded due to an inability to undergo brain MR imaging due to a neurostimulator in place. None were excluded for poor brain MR image quality. Thus, the final cohort was composed of 174 patients. One hundred thirteen (64.9%) were women, with average age 52.0 (SD, 14.3) years. At least 1 CSF leak was found on DSM in 76 (43.7%) patients, while indeterminate findings were noted in 22 (12.6%). Because indeterminate findings were considered suspicious and treated in all except 1 patient (21/22; 95.5%), such cases were considered positive. Thus, at least 1 CSF leak was discovered in 98 (56.3%) patients. There was no significant difference in sex makeup between patients without and with CSF leaks (P = .91).
For the entire cohort, the mean Dobrocky et al10 score was 4.0 (SD, 2.7). The average Dobrocky et al score among patients with a CSF leak (5.0) was significantly higher than in those without a CSF leak (2.6) (P < .001).
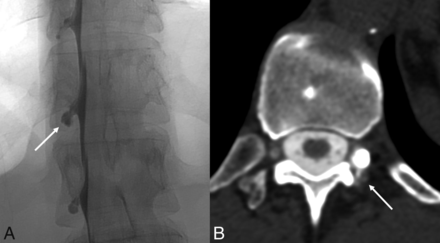
Regarding leak sites, 61/98 (62.2%) were on the right side, 29 (29.6%) were on the left side, 6 (6.1%) were bilateral, 1 (1.0%) was ventral, and 1 (1.0%) was into the internal epidural venous plexus. Ninety-three (94.9%) were classified as type 3 leaks (CSF-venous fistulas), 1 (2.0%) was type 1 (ventral dural leak), and 4 (4.1%) were type 4 (distal nerve root sleeve tears) (Fig 1).
An example of a distal nerve root sleeve tear (type 4 leak) diagnosed using a combination of DSM and delayed CTM findings. Left lateral decubitus unsubtracted image from a DSM (A) demonstrates contained contrast within a left T12 meningeal diverticulum (A, arrow). On a 30-minute delayed left decubitus CTM (B), there is a subtle contrast leak posterior to the diverticulum (B, arrow). The combined findings from a DSM and delayed CTM are often necessary to confidently diagnose type 4 leaks.
Brain Imaging
The average time between pre-DSM MR imaging and DSM was 91.0 (SD, 163.1) days. Eight patients did not have postcontrast imaging available for review.
Among the entire cohort, smooth dural enhancement was present in 63/166 patients (38.0%) and dural enhancement in the IACs was present in 38/166 (23.0%) (Fig 1). Unilateral or bilateral subdural fluid collections were observed in 13 (7.5%) patients. Superficial siderosis was present in 4 (2.3%) patients, though 1 of the 4 with superficial siderosis had a known history of prior head and spine trauma. Pituitary engorgement was observed in 73 (42.0%); non-Chiari cerebellar tonsillar descent, in 30 (17.2%); descent of the cerebral aqueduct iter below the incisural line, in 48 (27.6%); and layered hyperostosis, in 11 (6.3%). Venous sinus engorgement was clearly present in 30 (17.2%) patients and was indeterminate in 31 (17.8%). The average mamillopontine distance was 5.6 (SD, 1.9) mm, the average prepontine cistern width was 4.1 (SD, 1.5) mm, and the average suprasellar cistern height was 3.9 (SD, 2.5) mm.
Comparisons between Cohorts
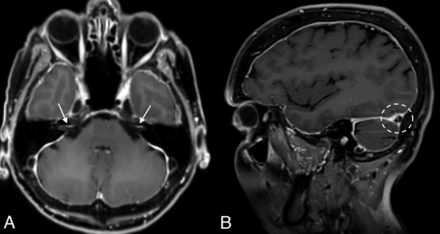
Comparisons of brain MR imaging findings in patients with and without a CSF leak identified on DSM are detailed in Table 1. Abnormalities significantly more common in patients with a CSF leak were smooth dural enhancement, dural enhancement involving the IACs, non-Chiari cerebellar descent of >5 mm below the foramen magnum, pituitary engorgement, dural venous sinus engorgement, descent of the cerebral aqueduct iter below the incisural line, and decreased suprasellar cistern height (Fig 2). Although subdural fluid collections, layered hyperostosis, and superficial siderosis were more common among patients with a CSF leak, the differences did not reach statistical significance. Similarly, the average mamillopontine distance and prepontine cistern width were smaller in patients with a CSF leak, though these differences did not reach statistical significance.
An example of multiple intracranial sequelae of SIH in a 69-year-old woman. Axial (A) and sagittal (B) postcontrast images demonstrate diffuse smooth dural enhancement, with involvement of the IACs (arrows, A). The right transverse sinus is engorged (dashed circle, B).
Frequency and measurements of intracranial findingsa
Dural venous sinus engorgement was associated with a CSF leak when allowing for categorization into 1) present, 2) absent, or 3) indeterminate classifications (P < .001). To incorporate this finding into the statistical analysis for the scoring system, we combined present and indeterminate into 1 category. By means of this method, the presence of definite or indeterminate dural venous sinus engorgement was still significantly associated with a CSF leak (P < .001).
By means of receiver operating characteristic analysis, the cutoff for the suprasellar cistern was 2.5 mm. A follow-up χ2 analysis assessing a suprasellar cistern size of ≤2.5 mm found that this size was significantly associated with the presence of a CSF leak (OR = 3.2; 95% CI, 1.6–6.3; P = .0008).
Development of Scoring System
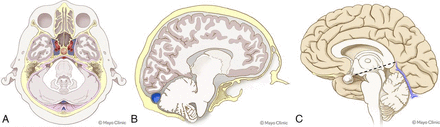
All statistically significant variables were given 1 point for the scoring system (Tables 2 and 3 and Fig 3). The average score for all patients was 2.1 (SD, 0.2) (range, 0–7). Higher scores were significantly associated with a CSF leak (OR = 1.7; 95% CI, 1.4–2.2; P < .001).
Schematics of intracranial findings used for the probabilistic scoring system developed in this study. Pertinent findings included diffuse dural enhancement and dural enhancement involving the walls of the IACs (pink lines, A), engorgement of the transverse sinus and non-Chiari cerebellar descent (B), and pituitary engorgement, effacement of the suprasellar cistern, and descent of the aqueduct iter below the incisural line (dotted line, C). Used with permission of Mayo Foundation for Medical Education and Research, all rights reserved.
Scoring system based on 7 intracranial imaging findings
Proposed probabilistic scoring system for determining whether a spinal CSF leak is present
Statistical analysis demonstrated the optimal cut-point for a probabilistic scoring system to be 3. No other cut-points were identified. Thus, our scoring system dichotomized the outcomes into “low” and “intermediate-to-high” probabilities of a CSF leak, divided into scores of 0–2 and ≥3, respectively. By means of this system, 65 patients were considered to have an intermediate-to-high probability of a CSF leak. Forty-three of 109 (39.4%) patients with a low-probability had a leak, while 55/65 (84.6%) patients with intermediate-to-high probability of a leak were found to have a leak.
The scoring system was significantly associated with finding a leak (OR = 8.4; 95% CI, 3.9–18.3; P < .001). By means of the scoring system of Dobrocky et al,10 both intermediate (OR = 1.4; 95% CI, 0.6–3.3) and high (OR = 8.3; 95% CI, 3.8–19.1) scores were associated with leaks (P < .001). There was no significant difference in the AUC for the scoring system of Dobrocky et al (AUC = 0.72) and the one developed for the current study (AUC = 0.70; P = .51).
DISCUSSION
We set out to create a new probabilistic scoring method for determining the likelihood of finding a spinal CSF leak in a patient with clinically suspected SIH. The results found that none of the “minor” criteria of the Dobrocky et al10 score were associated with a leak, while 7 of the studied intracranial findings were associated with a leak. From these, we developed a scoring system that dichotomized patients as having a low or intermediate-to-high probability of a CSF leak.
However, there was no significant difference between the 2 scoring systems in their ability to predict the presence or absence of a CSF leak, likely, due to a few major reasons. First, many of the same imaging findings are used by both systems. For example, our system uses the descent of the iter below the incisural line as a stand-in for subjective brain sag used previously. Next, many of the imaging findings coexist in patients with CSF leaks. Finally, the original study by Dobrocky et al10 had many strengths, including the use of statistical rigor and incorporation of a validation cohort to confirm its findings.
What then should be done with the 2 scoring systems: the one developed by Dobrocky et al10 and the scoring system developed in the current study? Ultimately, we believe that the scoring system developed in this study has some benefits over the prior system: It has less dependence on the measurement of minute structures and does not include the 3 minor findings that were not found to be associated with leaks. Nevertheless, the imperfections of both scoring systems highlight the concept that CSF leaks cannot be ruled in or out on the basis of imaging alone.
Next, the results of this study ultimately do serve as further validation of the Dobrocky et al10 system. Already, the Dobrocky system has been validated using both DSMs and dynamic CTMs.16,17 Therefore, the results of the current study, though they seek to reshape our assessment of intracranial findings in patients with suspected SIH, should also be thought to highlight the strengths of the Dobrocky et al scoring system.
The results of the current study should be interpreted in the context of its methodology. For example, superficial siderosis is commonly associated with fast CSF leaks, thought to be related to slow oozing of blood at the site of the dural defect.18 This superficial siderosis tends to be infratentorial and can regress after repair of the CSF leak.19 Prior reports have suggested that infratentorial siderosis is present in up to 20% of patients with a ventral spinal CSF leak.18 Because the current study excluded patients with an SLEC (and therefore a fast CSF leak), superficial siderosis was infrequently observed. The results of the current study should not be incorrectly interpreted to suggest that siderosis is not associated with spinal CSF leaks in general. Similarly, although subdural fluid collections did not reach the statistical threshold of being associated with a CSF leak in this study, these findings should not be misinterpreted to suggest that subdural collections are unrelated to SIH.
Finally, although nearly 40% of patients with a low probability of a score were found to have a leak, there is a substantial selection bias in the patients included in this cohort, all of whom had clinically suspected SIH. Nevertheless, given this large incidence of patients with relatively normal intracranial findings, the results of this study might suggest that DSMs may be warranted for patients with a high clinical suspicion of SIH.
This study has several limitations. First, the conclusions are based on a retrospective review. Prospective studies are needed to validate our results. In addition, because each patient was only reviewed by a single neuroradiologist, this study was not able to provide interobserver agreement analyses. Next, 2 findings were allowed to be given an “indeterminate” score during the initial assessment: whether a leak was visualized on DSM and/or the subsequent CT, and the presence or absence of dural venous sinus engorgement. In both cases, we lumped indeterminate findings into “positive” categories to best match the clinical realities. Also, there was some selection bias intrinsic to the methodology of this study, given that only patients without an SLEC were included. In addition, the reviewers were not blinded to the official reports or annotations in the PACS system. Finally, the statistical analysis of our data allowed the development of only a dichotomized scoring system, rather than the prior 3-tiered (low, indeterminate, high) system. For the clinician, however, this may actually simplify the decision to proceed or not to myelography.
CONCLUSIONS
This study set out to determine the likelihood of having a CSF leak based on intracranial MR imaging findings in patients without an SLEC. A new scoring system was developed on the basis of 7 intracranial findings associated with a leak. Some of the criteria used by the established scoring system did not meet the statistical threshold to include them in this new scoring system. Although this system was not significantly more accurate at predicting leaks than the Dobrocky method, it has potential benefits that could make it worthy of future studies.
Footnotes
Disclosure forms provided by the authors are available with the full text and PDF of this article at www.ajnr.org.
References
- Received June 20, 2023.
- Accepted after revision September 14, 2023.
- © 2023 by American Journal of Neuroradiology